Perpetual
Regulatory Compliance,
Simplified for Success
Designed for Every Regulation .
Mapped Workflows for Each Clause of QMS Standards and Regulations
QMS Standards:
- ISO 13485:2016
- EN ISO 13485:2016
- IEC 62304
- IEC 62366
- ISO 14971:2019; EN ISO 14971:2019
- US FDA 21 CFR 820
- Fifth Schedule of Indian MDR 2017
- MDSAP
Regulations:
- I-MDR 2017
- EU MDR 2017/745
- US FDA - 510(k) and Premarket submission

Mapped Workflows for Each Clause of QMS Standards and Regulations

QMS Standards:
- ISO 13485:2016
- EN ISO 13485:2016
- IEC 62304
- IEC 62366
- ISO 14971:2019; EN ISO 14971:2019
- US FDA 21 CFR 820
- Fifth Schedule of Indian MDR 2017
- MDSAP
Regulations:
- I-MDR 2017
- EU MDR 2017/745
- US FDA - 510(k) and Premarket submission
Meet Your Cloud-based eQMS Guide
With MedStrat360, companies can:
- Automation of all QMS processes including documentation, CAPA, audits, complaint handling, regulatory submissions, and more.
- Seamlessly adapt to global regulations like I-MDR, USFDA, EU-MDR, and more.
- Ensure traceability and accountability across the lifecycle of a medical device.
- Stay ahead with real-time system-wide updates in line with regulatory changes, enabling perpetual compliance.

The MeDStrat Innovations Difference
Although pre-market, MeDStrat Innovations is designed with measurable efficiency and compliance gains
in mind. These assumptions are based on industry benchmarks and the capabilities of our platform.
Due to streamlined regulatory submission processes.
Build Your Perfect Compliance Framework
- Document Management: Centralize, organize, and control documents with version tracking and approval workflows.
- CAPA (Corrective and Preventive Actions): Proactively manage quality issues with actionable CAPA workflows.
- Audit Management: Simplify internal and external audits with automated workflows and reporting.
- Competency and Training: Track employee competence and ensure continuous skill development.
- Complaint Management: Streamline the complaint management process to track, manage, and resolve issues efficiently.
- Management Review: Facilitate top-level review processes with actionable insights.
- Infrastructure Management: Optimize and oversee infrastructure to meet QMS and Regulatory requirements seamlessly.
- Data Analysis: Gain actionable insights through advanced analytics for decision-making.
- Control of Monitoring and Measurement Equipment: Ensure accurate and compliant equipment calibration and maintenance.

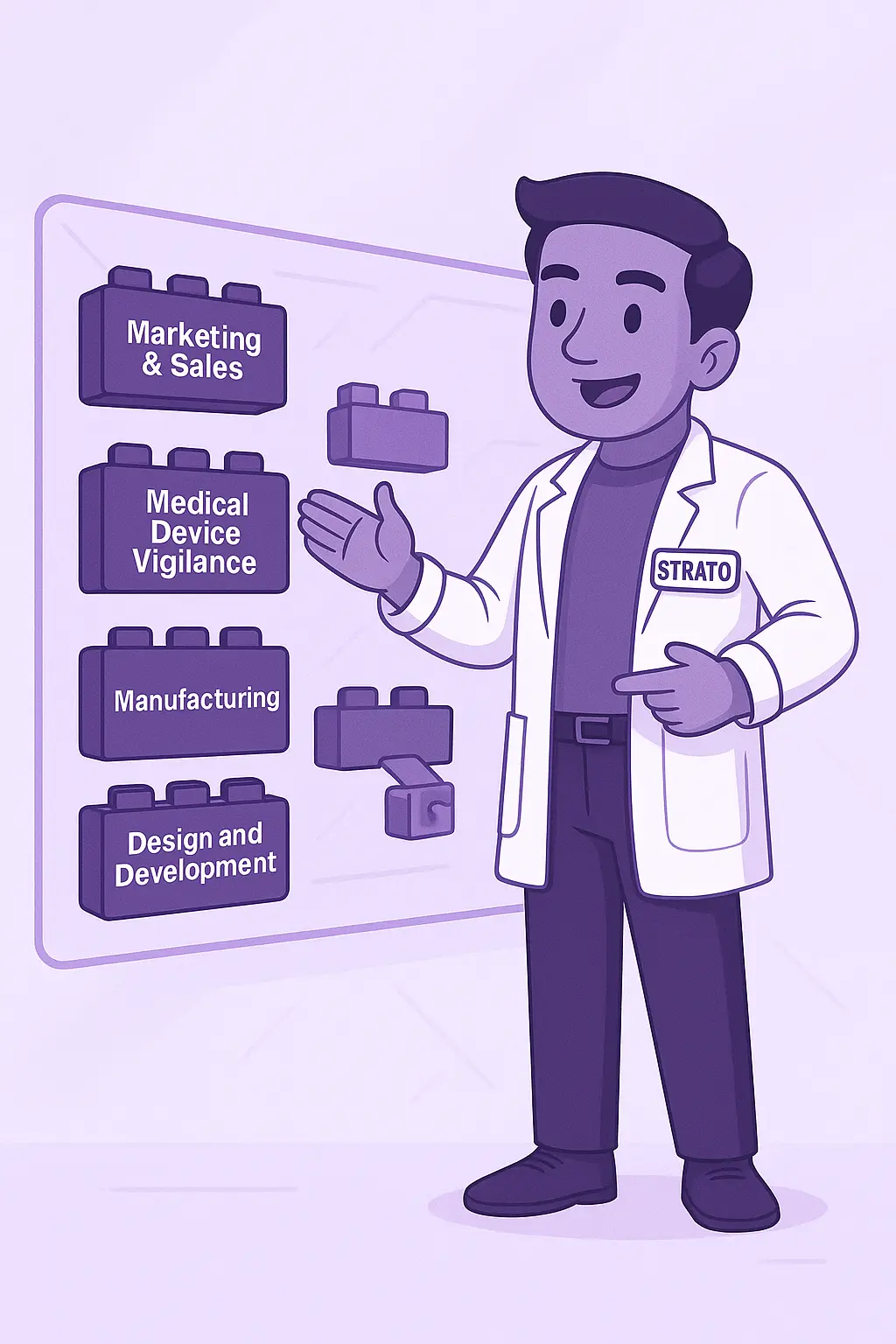
- Design and Development: Manage design processes, iterations, and approvals with traceability.
- Risk Management: Identify and mitigate potential risks across product lifecycles.
- Supplier and Outsourcing Management: Ensure supplier quality and manage outsourcing partnerships effectively.
- Medical Device Vigilance: Monitor, report, and follow up on real-world device performance with built-in workflows that support post-market surveillance and clinical follow-up compliance.
- Marketing and Sales: Track customer insights, manage promotional material approvals, and maintain compliance across regions with structured workflows that align with regulatory expectations for market communication.
- Manufacturing: Ensure product consistency and regulatory compliance by linking production processes with quality controls, traceability, and deviation management in one centralized system.
- Regulatory Submissions: Meet compliance requirements across multiple regions with efficient, system-driven regulatory submission processes.
Guiding Every Device to Regulatory Success
Pacemakers, infusion pumps, X-ray machines, and more

Surgical instruments, bandages, orthopaedic implants, and more.

Blood glucose monitors, COVID-19 test kits, pregnancy test kits, and more.

Software as a Medical Device (SaMD)
Mobile apps for diagnosing conditions, software for managing chronic diseases, AI-based radiology tools, and more.
Meet The Minds Behind MedStrat360
Sriram
CEO & Co-Founder
A compliance expert with over 40 years of experience in guiding medical device companies. Sriram’s strategic leadership ensures that MeDStrat Innovations remains at the forefront of regulatory advancements.
Kannan
CFO & Co-Founder
A seasoned entrepreneur with a rich background in finance and operations. Kannan brings business acumen and financial expertise to drive MeDStrat Innovation’s growth.
Ragav Kannan
Head of Marketing
A marketing strategist with global Operations experience at Apple, Caterpillar, and Lam Research, Ragav specializes in aligning market needs with scalable, compliance-driven solutions.

Contact Us
We’re always happy to help businesses reach their full potential with our tech expertise. Please let us know what service you are interested in by completing the form. We will get in touch with you shortly.